Technology Overview
Our team is dedicated to helping people survive cancer. Our passion is focused on enabling the medical community to achieve dramatic improvements in patient survival rates through the early detection of the leading forms of cancer.
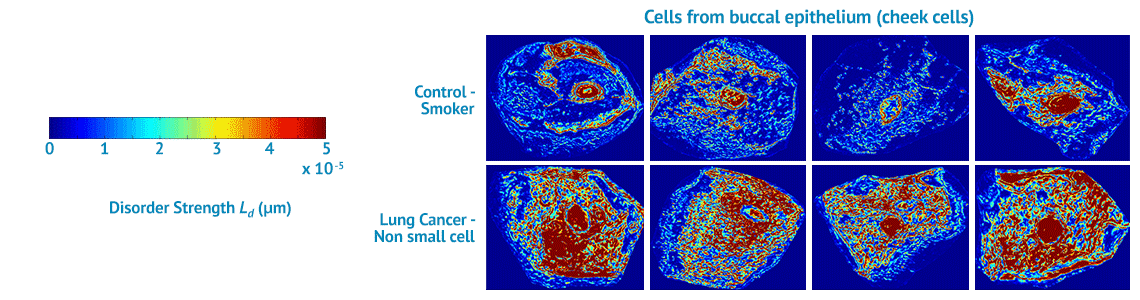
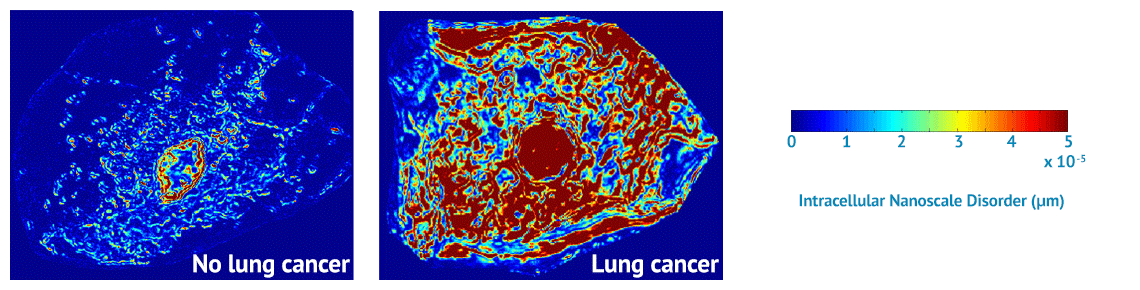
NanoCytomics’ contribution to this effort will be our cancer risk-stratification tests based on our novel, proprietary biophotonics technology platform known as partial wave spectroscopic (PWS) microscopy. Biophotonics deploys optical imaging and sensing technologies to study the structures and functions of cells. Through biophotonics in general and the company’s PWS platform in particular, NanoCytomics can identify cellular abnormalities at the nanoscale level. In so doing, the company anticipates making a profound impact on the ability to stratify cancer risk for an individual patient at a dramatically earlier stage of disease progression, potentially helping physicians to save millions of lives in the process.
NanoCytomics has an exclusive license from Northwestern University for patents and patent applications pertaining to the NanoCytomics technology and methods that were developed in Vadim Backman’s Northwestern Biophotonics Laboratory.